Abstract
In recent decades, intensive research on non-avian dinosaurs has strongly suggested that these animals were restricted to terrestrial environments1. Historical proposals that some groups, such as sauropods and hadrosaurs, lived in aquatic environments2,3 were abandoned decades ago4,5,6. It has recently been argued that at least some of the spinosaurids—an unusual group of large-bodied theropods of the Cretaceous era—were semi-aquatic7,8, but this idea has been challenged on anatomical, biomechanical and taphonomic grounds, and remains controversial9,10,11. Here we present unambiguous evidence for an aquatic propulsive structure in a dinosaur, the giant theropod Spinosaurus aegyptiacus7,12. This dinosaur has a tail with an unexpected and unique shape that consists of extremely tall neural spines and elongate chevrons, which forms a large, flexible fin-like organ capable of extensive lateral excursion. Using a robotic flapping apparatus to measure undulatory forces in physical models of different tail shapes, we show that the tail shape of Spinosaurus produces greater thrust and efficiency in water than the tail shapes of terrestrial dinosaurs and that these measures of performance are more comparable to those of extant aquatic vertebrates that use vertically expanded tails to generate forward propulsion while swimming. These results are consistent with the suite of adaptations for an aquatic lifestyle and piscivorous diet that have previously been documented for Spinosaurus7,13,14. Although developed to a lesser degree, aquatic adaptations are also found in other members of the spinosaurid clade15,16, which had a near-global distribution and a stratigraphic range of more than 50 million years14, pointing to a substantial invasion of aquatic environments by dinosaurs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available in the paper and its Supplementary Information. Three-dimensional data are available on SketchFab: flesh model at https://sketchfab.com/3d-models/07b2b6bf4c464c09bd30daa629f266ff; scanned caudal vertebrae and chevrons at https://sketchfab.com/3d-models/chv-7-ca70592e5d07408980220d639bc1456f (Chv7), https://sketchfab.com/3d-models/chv-24-d917f541a7934492aaf0be7c5b97ad40 (Chv24), https://sketchfab.com/3d-models/ca-4-56e19d32f53043369ba23d5283279eef (Ca4), https://sketchfab.com/3d-models/ca-7-c2f551b61e294138954c4f2224bd3353 (Ca7), https://sketchfab.com/3d-models/ca-12-bb2f30fec9064645b2ff99be30f1ac92 (Ca12), https://sketchfab.com/3d-models/ca-16-3cd5f6713e1f43f5bf466471416f06c8 (Ca16), https://sketchfab.com/3d-models/ca-23-f34b71eaf4e54cb29a89a0f50730e70a (Ca23), https://sketchfab.com/3d-models/ca-24-4e21ecea81c8403484b9b7e3da7bd0d6 (Ca24), https://sketchfab.com/3d-models/ca-31-6d0748a851994d6d86b84803743b75a4 (Ca31) and https://sketchfab.com/3d-models/ca-41-34e5e6415f7d4fcea335e7120b9fe9b4 (Ca41).
References
Weishampel, D. B., Dodson, P. & Osmólska, H. The Dinosauria 2nd edn (Univ. of California Press, Berkeley, 2004).
Owen, R. A description of a portion of the skeleton of the Cetiosaurus, a gigantic extinct saurian reptile occurring in the oolitic formations of different portions of England. Proc. Geol. Soc. Lond. 3, 457–462 (1841).
Cope, E. On the characters of the skull in the Hadrosauridae. Proc. Acad. Nat. Sci. Philadelphia 35, 97–107 (1883).
Kermack, K. A. A note on the habits of sauropods. Ann. Mag. Nat. Hist. 4, 830–832 (1951).
Bakker, R. T. Ecology of the brontosaurs. Nature 229, 172–174 (1971).
Alexander, R. M. Mechanics of posture and gait of some large dinosaurs. Zool. J. Linn. Soc. 83, 1–25 (1985).
Ibrahim, N. et al. Semiaquatic adaptations in a giant predatory dinosaur. Science 345, 1613–1616 (2014).
Aureliano, T. et al. Semi-aquatic adaptations in a spinosaur from the Lower Cretaceous of Brazil. Cretac. Res. 90, 283–295 (2018).
Evers, S. W., Rauhut, O. W. M., Milner, A. C., McFeeters, B. & Allain, R. A reappraisal of the morphology and systematic position of the theropod dinosaur Sigilmassasaurus from the “middle” Cretaceous of Morocco. PeerJ 3, e1323 (2015).
Henderson, D. M. A buoyancy, balance and stability challenge to the hypothesis of a semi-aquatic Spinosaurus Stromer, 1915 (Dinosauria: Theropoda). PeerJ 6, e5409 (2018).
Hone, D. W. E. & Holtz, T. R. Jr. Comment on: Aquatic adaptation in the skull of carnivorous dinosaurs (Theropoda: Spinosauridae) and the evolution of aquatic habits in spinosaurids. 93: 275–284. Cretac. Res. https://doi.org/10.1016/j.cretres.2019.05.010 (2019).
Stromer, E. Ergebnisse der Forschungsreisen Prof. E. Stromers in den Wüsten Ägyptens. II. Wirbeltier-Reste der Baharîje -Stufe (unterstes Cenoman). 3. Das Original des Theropoden Spinosaurus aegyptiacus nov. gen., nov. spec. Abh. Kgl. Bayer. Akad. Wiss. Math. Phys. Kl. München 28, 1–28 (1915).
Vullo, R. et al. Convergent evolution of jaws between spinosaurid dinosaurs and Pike Conger Eels. Acta Palaeontol. Pol. 61, 825–829 (2016).
Arden, T. M. S., Klein, C. G., Zouhri, S. & Longrich, N. R. Aquatic adaptation in the skull of carnivorous dinosaurs (Theropoda: Spinosauridae) and the evolution of aquatic habits in Spinosaurus. Cretac. Res. 93, 275–284 (2019).
Charig, A. J. & Milner, A. C. Baryonyx walkeri, a fish-eating dinosaur from the Wealden of Surrey. Bull. Nat. Hist. Mus. Geol. 53, 11–70 (1997).
Sues, H. D., Frey, E., Martill, D. M. & Scott, D. M. Irritator challengeri, a spinosaurid (Dinosauria: Theropoda) from the Lower Cretaceous of Brazil. J. Vert. Pal. 22, 535–547 (2002).
Witmer, L. M. in Mesozoic Birds: Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 3–30 (Univ. California Press, 2002).
Tereschenko, V. Adaptive features of protoceratopoids (Ornithischia, Neoceratopsia). Paleontol. J. 42, 273–286 (2008).
Cau, A. et al. Synchrotron scanning reveals amphibious ecomorphology in a new clade of bird-like dinosaurs. Nature 552, 395–399 (2017).
Sereno, P. C. et al. A long-snouted predatory dinosaur from Africa and the evolution of spinosaurids. Science 282, 1298–1302 (1998).
Lavocat, R. Sur les dinosauriens du Continental Intercalaire des Kem-Kem de la Daour. In Comptes rendus 19e Congrès Géologique International, Alger, 1952 65–68 (Academie des Sciences de Paris, 1954).
Sereno, P. C. et al. Predatory dinosaurs from the Sahara and Late Cretaceous faunal differentiation. Science 272, 986–991 (1996).
D’Août, K. & Aerts, P. Kinematic and swimming efficiency of steady swimming in adult axolotls (Ambystoma mexicanum). J. Exp. Biol. 200, 1863–1871 (1997).
Fish, F. Kinematics of undulatory swimming in the American alligator. Copeia 1984, 839–843 (1984).
Frolich, L. M. & Biewener, A. A. Kinematic and electromyographic analysis of the functional role of the body axis during terrestrial and aquatic locomotion in the salamander Ambystoma tigrinum. J. Exp. Biol. 162, 107–130 (1992).
Lauder, G. V., Flammang, B. & Alben, S. Passive robotic models of propulsion by the bodies and caudal fins of fish. Integr. Comp. Biol. 52, 576–587 (2012).
Cavin, L. et al. Vertebrate assemblages from the early Late Cretaceous of southeastern Morocco: an overview. J. Afr. Earth Sci. 57, 391–412 (2010).
Meunier, L. M. V. & Larsson, H. C. E. Revision and phylogenetic affinities of Elosuchus (Crocodyliformes). Zool. J. Linn. Soc. 179, 169–200 (2017).
Amiot, R. et al. Oxygen and carbon isotope compositions of middle Cretaceous vertebrates from North Africa and Brazil: ecological and environmental significance. Palaeogeogr. Palaeoclimatol. Palaeoecol. 297, 439–451 (2010).
Hone, D. W. E. & Holtz, T. R. A century of spinosaurs. A review and revision of the Spinosauridae with comments on their ecology. Acta Geol. Sin. 91, 1120–1132 (2017).
Ryan, M. J., Russell, A. P., Eberth, D. A. & Currie, P. J. The taphonomy of a Centrosaurus (Ornithischia: Ceratopsidae) bone bed from the Dinosaur Park Formation (Upper Campanian), Alberta, Canada, with comments on cranial ontogeny. Palaios 16, 482–506 (2001).
Erickson, G. M., Currie, P. J., Inouye, B. D. & Winn, A. A. Tyrannosaur life tables: an example of nonavian dinosaur population biology. Science 313, 213–217 (2006).
Bertozzo, F., Dalla Vecchia, F. M. & Fabbri, M. The Venice specimen of Ouranosaurus nigeriensis (Dinosauria, Ornithopoda). PeerJ 5, e3403 (2017).
Chinsamy, A. & Raath, M. A. Preparation of fossil bone for histological examination. Palaeontol. Afr. 29, 39–44 (1992).
Lee, A. H. & O’Connor, P. M. Bone histology confirms determinate growth and small body size in the noasaurid theropod Masiakasaurus knopfleri. J. Vertebr. Paleontol. 33, 865–876 (2013).
Horner, J. R. & Padian, K. Age and growth dynamics of Tyrannosaurus rex. Proc. R. Soc. Lond. B 271, 1875–1880 (2004).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Lauder, G. V., Anderson, E. J., Tangorra, J. & Madden, P. G. A. Fish biorobotics: kinematics and hydrodynamics of self-propulsion. J. Exp. Biol. 210, 2767–2780 (2007).
Lauder, G. V. et al. Robotic models for studying undulatory locomotion in fishes. Mar. Technol. Soc. J. 45, 41–55 (2011).
Quinn, D. B., Lauder, G. V. & Smits, A. J. Maximizing the efficiency of a flexible propulsor using experimental optimization. J. Fluid Mech. 767, 430–448 (2015).
Rosic, M. N., Thornycroft, P. J. M., Feilich, K. L., Lucas, K. N. & Lauder, G. V. Performance variation due to stiffness in a tuna-inspired flexible foil model. Bioinspir. Biomim. 12, 016011 (2017).
Saadat, M. et al. On the rules for aquatic locomotion. Phys. Rev. Fluids 2, 083102 (2017).
Read, D. A., Hover, F. S. & Triantafyllou, M. S. Forces on oscillating foils for propulsion and maneuvering. J. Fluids Structures 17, 163–183 (2003).
Shelton, R. M., Thornycroft, P. J. & Lauder, G. V. Undulatory locomotion of flexible foils as biomimetic models for understanding fish propulsion. J. Exp. Biol. 217, 2110–2120 (2014).
Acknowledgements
We thank M. Azroal, H. Azroal, M. Fouadassi and all other expedition members from the 2015, 2018 and 2019 seasons for assistance in the field; A. A. Ha for help in preparing the fossils; the Moroccan Ministry of Mines, Energy and Sustainable Development for providing fieldwork permits; F. Manucci for helpful discussions about the flesh reconstruction of Spinosaurus; and P. Fahn-Lai for coding assistance. This research was supported by a National Geographic Society grant to N.I. (CP-143R-170), a National Geographic Emerging Explorer Grant to N.I., contributions from the Board of Advisors of the University of Detroit Mercy to N.I., a Jurassic Foundation grant to M.F., a Paleontological Society grant to M.F., an Explorers Club grant to M.F., as well as financial support from the Lokschuppen Rosenheim, the Museo di Storia Naturale di Milano, J. Pfauntsch and A. Lania.
Author information
Authors and Affiliations
Contributions
N.I. led the expeditions and the project. N.I., S.M., C.D.S., M.F., M.A., D.M.M., G.B., S.Z., D.A.M. and A.A. collected the specimens in the field. N.I., S.M., C.D.S., M.F., J.W., G.V.L. and S.E.P. designed the research. N.I., S.M., C.D.S., M.F., J.W., G.V.L. and S.E.P. designed and performed the experiments. N.I., S.M., C.D.S., M.F., M.A., D.M.M., J.W., G.B., S.Z., D.A.M., D.M.U., U.J., J.J., A.A., G.V.L. and S.E.P. analysed the data. G.B., M.A., C.D.S., S.E.P., D.M.M., S.M., M.F. and D.B. created the figures. D.B. sculpted the life reconstruction. N.I., S.M., C.D.S., M.F., J.W., D.M.U., G.V.L. and S.E.P. wrote the manuscript, which was reviewed by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Matthew C. Lamanna, John A. Nyakatura and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
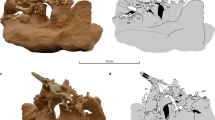
Extended Data Fig. 1 Excavation of the FSAC-KK 11888 site.
a–f, Different stages of the excavation, which resulted in the removal of over 15 tons of rock using a range of tools, including picks, brushes, hammers and a jackhammer. a, 17 November 2013. b, 29 March 2015. c, 17 September 2018. d, 19 September 2018. e, 5 December 2018. f, 21 July 2019. g, h, Selected bones in situ. g, Largely complete proximal caudal vertebra (Ca4). h, Neural spine of a mid-distal caudal vertebra, fragmented by syndiagenetic cracks. Scale bars, 10 cm.
Extended Data Fig. 2 Excavation of the caudal elements of FSAC-KK 11888.
a, Largely complete distal caudal vertebra (Ca31), recovered in its entirety by digging a tunnel until the apex of the neural spine was reached. b, Semi-articulated mid-caudal vertebrae. c, Two haemal arches. d, Close association of middle caudal elements. Scale bars, 10 cm.
Extended Data Fig. 3 Excavation map and skeletal reconstruction.
Detailed map of the site of discovery of FSAC-KK 11888, and fully revised skeletal reconstruction. Colours in the map and reconstruction correspond to different phases of excavation: the local discovery in 2007–2008 (red), our excavations during the 2015–2019 expeditions (green) and sieving in the debris area during the 2015–2019 expeditions (yellow). Both images are at the same scale. Scale bar, 1 m.
Extended Data Fig. 4 The caudal series of FSAC-KK 11888.
Photograph of the entire caudal series (numbered). Scale bar, 1 m.
Extended Data Fig. 5 Elements of FSAC-KK 11888 from 2008 (first excavation) and 2019 (most recent excavation), matched.
a–t, Evidence of the perfect match between elements collected by the local discoverer of the site in 2007–2008 (a, e, f, i, k, m, n, p, q, s) and elements excavated in situ or recovered from the site debris during the 2015–2019 excavations (b–d, g, h, j, l, o, r, t). a, b, Right and left metatarsal II. c, Left penultimate phalanx of the fourth pedal digit (IV-4) that came to light within the typical matrix in which bones of the FSAC-KK 11888 were embedded. d, e, Two possible splenial fragments reconnected. f–i, Phalanx IV-4 prepared and compared to the contralateral element of the right pes in dorsal view, and rearticulated with its ungual. j–m, Two complementary (broken) halves of the left squamosal and of a dorsal rib. n–p, s, t, Two key fragments from the debris, reconnecting the base and the shaft of a neural spine (possibly the 7th). q, r, The right astragalus (excavated in situ in July 2019) rearticulated to its tibia (from 2008). Arrows point to recomposed fractures.
Extended Data Fig. 6 Comparison of FSAC-KK 11888 caudal vertebrae to those destroyed in World War II.
Comparison between the caudal vertebrae of the neotype of S. aegyptiacus, with those of the two specimens (the holotype and a specimen known as ‘Spinosaurus B’, both of which are now lost) from the Bahariya Oasis (Egypt) described by E. Stromer12. a–d, Proximal caudal vertebra of the holotype (accession code BSP 1912 VIII 19) in distal (a) and right lateral (c) views; and of Ca4 of FSAC-KK 11888 in distal (b) and right lateral (d) views. e–j, Proximal caudal vertebra of Spinosaurus B in dorsal (e), right lateral (g) and proximal (i) views; Ca11 of FSAC-KK 11888 in dorsal (f), right lateral (h) and proximal (j) views. k–p, Middle caudal vertebra of Spinosaurus B in dorsal (k), left lateral (m) and distal (o) views; Ca21 of FSAC-KK 11888 in dorsal (l), left lateral (n) and distal (p) views. Scale bars = 10 cm.
Extended Data Fig. 7 Three-dimensional fleshed-out model based on FSAC-KK 11888.
a, b, Symmetrical pose in five views (a) and swimming pose (b).
Extended Data Fig. 8 Whole-body centre of mass.
a–d, Snout tip and tail tip in the coordinate system (a) and centre-of-mass distance from the cranial margin of the acetabulum in this study (b), calculated using multiple approaches (Supplementary Information, Supplementary Data 2) and compared to the centre of mass in ref. 10 (c); and ref. 7 (d).
Supplementary information
Supplementary Information
This file contains Supplementary Text (Parts 1-9) and Figures (S1-S4) related to: Geological context; Description of new material; Evidence for single individual; Referral to Spinosaurus aegyptiacus; Photogrammetry; Whole body reconstruction; Body mass and centre of mass; Anatomical abbreviations; and References.
Supplementary Data 1
Raw thrust and efficiency data, including mean and standard error.
Supplementary Data 2
Body dimensions, body mass, body segment masses, and whole body centre of mass.
Video 1
Excavation of Spinosaurus tail.
Video 2
Photogrammetry of caudal vertebra.
Video 3
Spinosaurus tail shape undulating in water flume from lateral perspective.
Video 4
Spinosaurus tail shape undulating in water flume from ventral perspective.
Video 5
Spinosaurus tail shape undulating in water flume from posterolateral perspective.
Rights and permissions
About this article
Cite this article
Ibrahim, N., Maganuco, S., Dal Sasso, C. et al. Tail-propelled aquatic locomotion in a theropod dinosaur. Nature 581, 67–70 (2020). https://doi.org/10.1038/s41586-020-2190-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2190-3
This article is cited by
-
Articular surface interactions distinguish dinosaurian locomotor joint poses
Nature Communications (2024)
-
Variability of spinosaurid teeth in the Barremian of the province of Teruel (eastern Spain)
Journal of Iberian Geology (2024)
-
A new spinosaurid dinosaur species from the Early Cretaceous of Cinctorres (Spain)
Scientific Reports (2023)
-
Growth constraints set an upper limit to theropod dinosaur body size
The Science of Nature (2023)
-
Subaqueous foraging among carnivorous dinosaurs
Nature (2022)